This the first tutorial on modeling heat transfer at a very introductory level.
If you follow this series and spend your own effort in developing your own models you will be able to model heat transfer in very complex shapes (1D, 2D, 3D) in a short time and with the basic understanding of a 12 year old school boy.
![]()
Animated heat transfer modeling for the average Joe – part #1
by George Lungu
-This is an introduction to heat transfer modeling. Up until today
I have not had much exposure to the subject except for the basic
thermodynamics I had in my first and second trimesters of the
10th grade (high school physics).
-Back in the day while studying electrical engineering in
Bucharest I disliked a subject called “The microwave
technology”. The class was heavy with math, notations and hours
of boring derivation to prove even something of small significance.
– With all of this in mind I will try to make the topic as easy and
intuitive as possible. Try opening a book on heat or read the page
in Wikipedia and you’ll probably ruin your day.
A 12 year old has the mental power and life experience to understand the base of
the heat transfer concepts, or electromagnetic field, or fluid
dynamics. That changes in college when our brain gets corrupted
with too much vector calculus, Greek alphabet and our confidence
in our own reason is forcibly shaken.
<excelunusual.com> by George Lungu
A rant: why sometimes standard teaching spoils your chances of learning?
– The answer is, it spoils motivation and it scares the
shy and curious part of our brain who is responsible for
creativity.
– I have seen the theory about heat transfer, or EM
fields or fluid dynamics and combined with complicated
notations and operators I think is perfect for preserving
the knowledge or advancing it at high level but not for
starting the learning process.
There is always a degree of understanding associated with literal derivation but usually deep
understanding comes from thinking the process over and over (and over again), playing, enjoying,
experimenting, and going to sleep thinking about it without notations/formulas/operators rather
than from just reading/writing/teaching/patenting.
-Beginning the process of learning with letters and derivations I believe is a counterproductive
proposition. It is unproductive because it gives you a surrogate of understanding.
Secondly, starting with literal derivations kills genuine curiosity and interest (how many students are in the class only
with their body – their mind is out).
There is a third danger witch comes from the fact that the
student, after passing the exam, has the illusion that he or she actually learned the matter.
– I also believe that the early steps in learning are more important than the later ones. Let’s
launch a rocket to Mars pointing slightly down and see what happens. But if the first few meters
are pointed in the right direction the rocket will probably do well for the next 1 billion miles.
How about some analogies?
– Most people understand electricity when they finally start thinking about electricity-water
analogy.
– The electricity-water analogy connects charge with amount of water, current intensity with
water flow, voltage difference with pressure difference, current flow through a resistor with the
impeded water flow through a narrow pipe, etc.
– The water-heat analogy will be connecting amount of water with heat, water flow with heat
flow, water pressure with temperature, a barrel as a water collector with a body as a heat
collector, water resistance or conductance due to pipe characteristics with heat resistance or
conductance due to the thermal bonding characteristics.
Electricity: Water: Heat:
1. Electric 1. Water 1. Heat
charge amount amount
2. Electric current 2. Water flow 2. Heat flow
3. Voltage 3. Pressure 3. Temperature
4. Resistance 4. Water flow 4. Heat
resistance resistance
Basic storage principles
– I can briefly enunciate them but it’s up to you to keep thinking about them for a while to see
if they really make sense. I like to keep it light and casual without being too rigorous as I
were to explain them to a 12 year old (IEEIKS = If Everything Else Is Kept the Same):
1. IEEIKS a body stores heat proportional to its mass.
2. IEEIKS a body stores heat in relation to the substance it is made from.
3. IEEIKS the net amount of heat a body exchanges with the environment
in a certain time interval is proportional to the net temperature
change the same body experiences during that time interval.
Let’s see if the heat storage principles make sense
Q > Q
1. A young pretty Aleutian girl, who felt cold in her sleep during
all her childhood, is getting married. Online she finds two options: a
large man and a skinny man who himself suffered from the same
problem in his own childhood. Whom will she choose?
– The problem is not rigorous since the human body is not just a
reservoir of heat but a generator too. She is happy because for her
the answer is simple, she will marry the bigger guy since he can store
(and produce) more heat, unless she is the psycho type who likes to – 27 year old, large,
well endowed
relate with her spouse in misery. leutian home boy…
2. Some substances store heat better than others. Hydrogen and also
water are known for storing the largest amount of heat per unit – 31 year old, very
well fit Eskimo man,
excellent hunter…
weight. Metals are poor at storage but good at conducting heat.
This second principle has been proven experimentally so there is not much to think about.
3. The third principle is intuitive too, just try to take a large sip from a cup of water at room
temperature (which is just 10 degrees cooler than the body) or from a cup of icy water (which
is 37 degrees cooler).
Before swallowing, try to measure how long it takes to roughly equalize
the temperature of the water in your mouth with the temperature of the body.
Three storage principles in one formula: Q c m
The previously mentioned principles are incorporated in the above formula. “Q” is the stored heat
variation [Joules], “m” is the mass of the object [Kg], “c” is called specific heat and it’s a material specific
property [J/Kg*K] and “T” is the temperature variation [K]. I’ve seen this formula both in middle school
and in high school. I’ve seen it in absolute form or in a differential form (like here). Numerical people can
model the moon in the sky and they almost always prefer things differential.
Often times for a given body, people like
to bundle “m” and “c” together to form
Q c m
Q C
C=c*m the thermal capacitance [J/K]
and in this case the formula becomes =>
Isn’t the previous formula for thermal
capacity quite similar to the formula
for the electrical capacity?
You can verify the analogy two pages back.
We can see even more similarities if we look a little farther to the standard formulas of thermal capacity and electric capacitance of a planar capacitor:
-Both specific heat and electric thermal permittivity are material constants
C cm
– Mass, area and dielectric thickness depend on the body geometry
C-electric
Conclusions:
In this part of the tutorial a few very basic heat concepts were introduced together
with the heat storage as an analogy to electric charge storage on a capacitor. The
next tutorial will deal with the concept of heat transfer or heat movement
between a body and its environment.
to be continued…
by George Lungu <excelunusual.com>

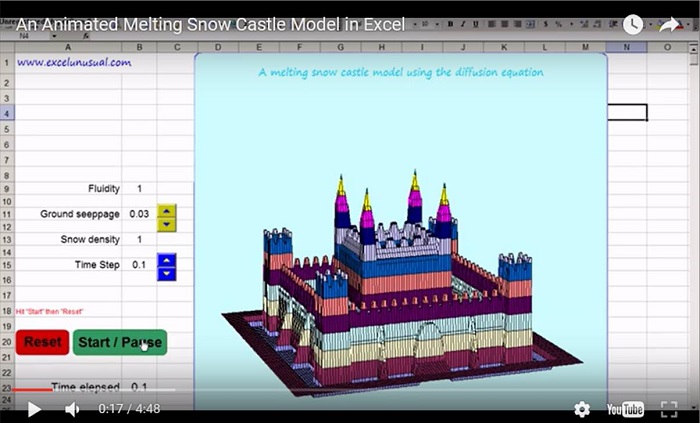
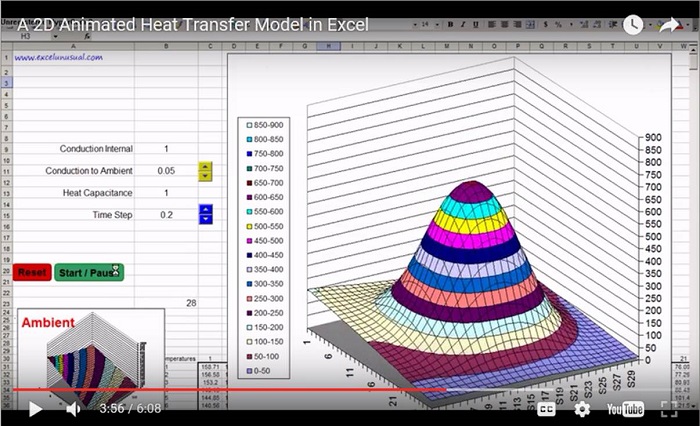
Can’t wait for #2.
Thx for the detailed explanation.
You’re welcome Niuxx. I have only 2 pages left but there is a Simpsons I need to watch first with my son. George
waiting for tutorial in detail
Today I am posting the first part. I got caught in details. Tomorrow or hopefully late tonight I will post the second part – how to program the thing.